Electric magnetic treatment device
Soken (Ver. 220V)
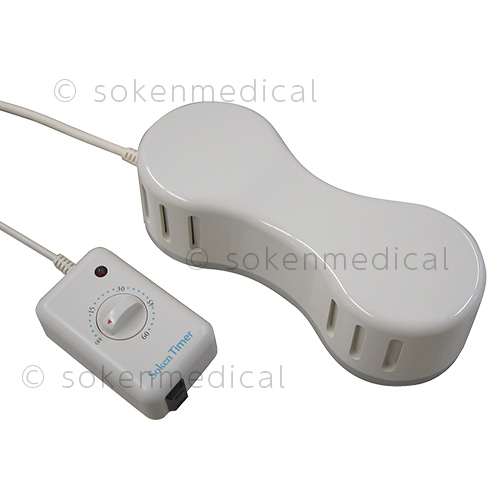
A long-selling magnetic shower by the trusted brand
It is an exclusive unit to use it abroad (Ver.220V).
| Brand name | SOKEN(Ver.220V) |
|---|---|
| Model | Ⅵ type(Ver.220V) |
| General name | Electric magnetic therapy device for household use |
| Device class | Controlled medical device |
| Authorization No. | 15500BZZ01533000 |
| Flux density on the surface | 80 mT (Bmax) 50 Hz 66 mT (Bmax) 60 Hz |
| Power supply | 220V 50Hz / 60Hz |
| Nominal capacity | 110 VA for 50 Hz 92 VA for 60 Hz |
| Safety system | Thermostat (60oC) and fuse (76oC) |
| Detentions/Weight (main unit) | 270 (W) x 73 (H) x 113 (D) mm/2.53 kg |
| Supplied timer | Max. 60 min (auto-OFF), |
| Hope retail price | Open |
Sokenrelax (Ver. 220V)

Top athletes also pay attention. Portable type that is convenient to carry
It is an exclusive unit to use it abroad (Ver.220V).
| Brand name | SOKEN RELAX(Ver.220V) |
|---|---|
| Classification | Mechanical Equipment 81 |
| Common Name | Electromagnetic Treatment Device(JMDN Code: 71015000) |
| Medical Device Type | Controlled medical device |
| Japan Medical Device Authorization | 22000BZX00235000 |
| Surface Inductive Flux | 38mT |
| Power Source/Capacity | AC220V50Hz / 0.22A/48.4VA±15% |
| Safety Device | Thermostat 80℃ Temperature fuse 115℃ |
| Timer | Max. 60 min. (Auto OFF) |
| Main Unit Dimensions | H 355xW 540xD 32(mm) |
| Weight | 2.65kg (Including accessories) |
| Hope retail price | Open |
Safety precautions
●Before you use this device, read the attached user manual thoroughly to ensure proper operation.
●Be sure to apply the supplied cover to the device. ●Make sure to use the device with the supplied timer.
Contraindications
Do not use this product with the following electrical medical devices:
(1) Implantable medical devices such as pacemakers
(2) Adjustable pressure shunt valves for cerebrospinal fluid shunting*
(3) Life-support equipment such as artificial heart-lung machines
(4) Electrical medical devices worn on the body, such as electrocardiograms
Use of these medical devices may cause an accident, possibly resulting in injury.
*An adjustable pressure valve is a medical device for shunting treatment that is intended to channel the cerebrospinal fluid into the peritoneal cavity. The valve is implanted under the scalp for a long period to adjust the flow volume of the cerebrospinal fluid by changing the valve pressure with magnetic energy.
Cautions
If you have any of the following conditions, consult your physician before using this product:
●Malignant tumors ●Heart problems ●In the unstable period of the early stages of pregnancy or immediately after delivering ●Perceptual disorders due to major peripheral circulatory disturbance caused by diabetes mellitus ●Loss of temperature sensation ●Skin infections/wounds ●A condition that requires rest ●Having a temperature of 38ºC or higher ●Acute diseases such as distortion or muscle strain
When using the product, do not place it close to or bring it in contact with the following:
●Mobile phones ●watches ●cash cards ●hearing aids ●precision equipment ●PCs and peripheral devices (including disc drives)



